Ernest Rutherford: Father of Nuclear Physics
By Xiao P. Li
Ernest Rutherford, born on August 30, 1871, in Nelson, New Zealand, in a family of seven sons and five daughters, was the fourth child and second son. Rutherford went to public school like most of the kids today when he was little. Excelling in school, at the age of 16, Rutherford was able to obtain a scholarship to attend Nelson Collegiate School, a private all boys school. His brilliance in academics did not just end there. In 1889, Rutherford was awarded a University Scholarship allowing him to attend the University of New Zealand and later in Canter College. He graduated in 1893 with double first in Mathematics and Physics and he continued to research in his college after his graduation. The next year, Rutherford was able to receive the 1851 Exhibition Science Scholarship. Rutherford continued to receive awards and advance in the field of science later on in his life, but his greatest award of all is the Nobel Prize in Chemistry in 1908. This award was rewarded to him when he was acknowledged for his investigations into the disintegration of the elements, and the chemistry of radioactive substances in the new explored field, Nuclear Chemistry.
Rutherford may sound like a super human to us all with his achievements in life, but he is just like all of us. With passion and determination, one can also achieve such goal. I know I want to be like him and win a Nobel Prize in Chemistry.
Rutherford may sound like a super human to us all with his achievements in life, but he is just like all of us. With passion and determination, one can also achieve such goal. I know I want to be like him and win a Nobel Prize in Chemistry.
Some Achievements of Ernest Rutherford in Science:
-He discovered the concept of radioactive half-life in Nuclear Chemistry.
-He was able to differentiate between radioactive alpha and beta particles.
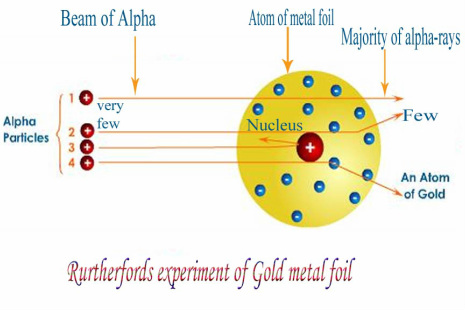
-With his Gold Foil Experiment, Rutherford theorized that atoms have their mass concentrated in the nucleus. He is correct because the majority of the mass of an atom, composing of protons and neutrons, are in the nucleus. Note that electron do have mass, but it is so small that it does not impact the overall mass of the atom. During the experiment, Rutherford shot alpha particles through a thin sheet of gold foil. Thinking that the alpha particle is so large, it should pass through the gold foil with ease. However, he observed that once in a while, alpha particles bounced back from the gold foil. This led to the conclusion of the atom having a dense nucleus and the atom is composed of mostly empty space.
-Also with the Gold Foil Experiment, Rutherford was able to discover and name the sub-atomic particle proton.
-With the help of James Chadwick in 1932, Rutherford and Chadwick was able to discover and name the sub-atomic particle neutron.
-Rutherford was able to perform the first splitting of an atom in 1917 between nitrogen and alpha particles. This experiment helped contribute to the development of the Nuclear Bomb during World War II.
-He was able to differentiate between radioactive alpha and beta particles.
-With his Gold Foil Experiment, Rutherford theorized that atoms have their mass concentrated in the nucleus. He is correct because the majority of the mass of an atom, composing of protons and neutrons, are in the nucleus. Note that electron do have mass, but it is so small that it does not impact the overall mass of the atom. During the experiment, Rutherford shot alpha particles through a thin sheet of gold foil. Thinking that the alpha particle is so large, it should pass through the gold foil with ease. However, he observed that once in a while, alpha particles bounced back from the gold foil. This led to the conclusion of the atom having a dense nucleus and the atom is composed of mostly empty space.
-Also with the Gold Foil Experiment, Rutherford was able to discover and name the sub-atomic particle proton.
-With the help of James Chadwick in 1932, Rutherford and Chadwick was able to discover and name the sub-atomic particle neutron.
-Rutherford was able to perform the first splitting of an atom in 1917 between nitrogen and alpha particles. This experiment helped contribute to the development of the Nuclear Bomb during World War II.